Aquabrid™
Surgical Sealant
Developed for wet conditions
Aquabrid is a fully synthetic surgical sealant for aortic surgical procedures. In contact with water Aquabrid forms an elastic layer within 3 to 5 minutes – making it optimal for use on wet conditions.1,2
Aquabrid stretches and shrinks with contraction of the vessel, while maintaining a strong seal in the aorta.1,2 Aquabrid has been commercially available in Japan since 2014 under the name of HYDROFIT®.
Benefits of Aquabrid

Reacts with water1,2
Optimal use for wet surfaces, regardless of heparinization conditions

Ready to use3
No manual mixing or preparation required
Elastic1,2
Stretches and shrinks with the vessel contractions

Strong1,2
Maintains bond/seal in the high pressure environment of the aorta

100% synthetic4,5
No biological origin or risk of infection
How does Aquabrid work?
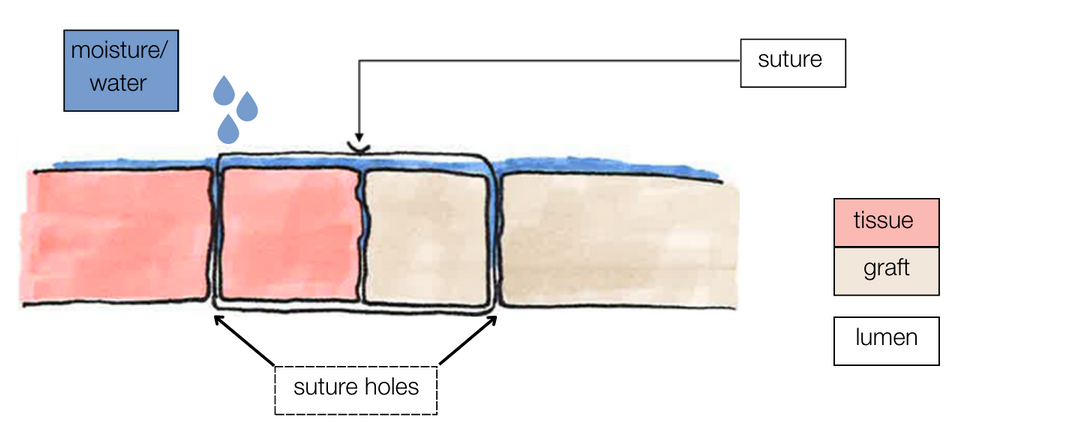
Aquabrid can be applied to natural and/or artificial tissue (e.g. aortic graft). The surgical site does not need to be dry, Aquabrid requires blood/moisture for reaction.
A thin layer of Aquabrid (liquid) is applied to seal the suture holes.
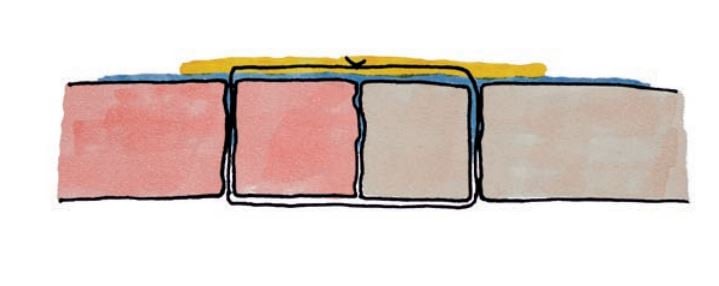
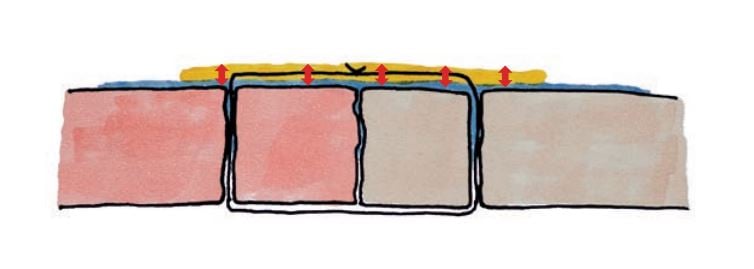
Aquabrid absorbs the moisture/blood and doubles it´s regional volume.
Aquabrid forms an elastic film after polymerization with CO2 release.
Application of Aquabrid
Direct Method
Transfer Method
Discover aortic surgical procedures with Aquabrid

Abdominal infrarenal aoritc aneurysm repair
A thin layer of Aqaubrid supports the seal of aortic anastomosis during abdominal infrarenal aoritc aneurysm repair.

Graft prothesis anastomosis
Aquabrid stops bleeding from suture holes during graft prothesis anastomosis.

Bentall procedure
Aquabrid stops bleeding from all anastomosis points during aortic arch replacement.

Aortic arch replacement
Aquabrid stops bleeding from anastomosis at heart side, even though difficult to access with silicone sheet.
.png?width=1200&length=1200&name=Frozen%20elephant%20trunk%20(FET).png)
Frozen elephant trunk (FET)
Aquabrid stops bleeding from anastomosis of graft and vessel.
 Did we spark your interest?
Did we spark your interest?
Book a virtual training or request an in-house ‘hands-on’ session.
Access our large variety of learning resources, such as narrated live case videos, webinars, eLearnings etc. and take a deep dive into the Aquabrid application methods, features and benefits of the product, tips & tricks from experienced peers ...
and much more!
REFERENCES.
1. Eto M et al. Elastomeric surgical sealant for hemostasis of cardiovascular anastomosis under full heparinization. Europ. J. Cardio Surg. 2007; 730-734.
2. Oda S. et al. Experimental use of an elastomeric surgical sealant for arterial hemostasis and its long-term tissue response. Interac. Cardiov. and Thor. Surgery. 2010; 258-261.
3. Instruction for use.
4. Annabi N et.al. 2015: Elastic sealants for surgical applications; Eur J Pharm Biopharm; 95(0 0): 27-39.
5. Lodi D et. al. 2012: Management of hemostasis in surgery: sealant and glueapplications; Blood Coagulation and Fibrinolysis; 2012, Vol. 23, No. 6, 465-472.
 Designed and manufactured by Sanyo Chemical Industries, LTD.
Designed and manufactured by Sanyo Chemical Industries, LTD.
11-1, Ikkyo Nomoto-cho, Higashiyama-ku, Kyoto 605-0995, Japan

